|
Therefore, the mass percentage of the OH in Ca(OH)₂ is equal to 45.94%. sulfate is SO 4 2-, so sulfite has the same charge but one less oxygen (SO 3 2-) b. An atom of oxygen with a negative charge contains. If you memorize the ate ions, then you should be able to derive the formula for the ite ion and vice-versa. , Electron affinities from endothermic negative ion charge transfer reactions. Because an oxygen atom with zero charges contains six electrons, the oxidation state of the atom is zero. Note that there are two polyatomic ions in this table, the ammonium ion and hydronium ion that are cations. ate anions have one more oxygen then the ite ion, but the same charge. For example, NO3 NO 3 is the nitrate ion it has one nitrogen atom and three oxygen atoms and an overall 1 charge. The molecular mass of the Ca(OH)₂ = 74 g/mol 1 lists the ion names and ion formulas of the most common polyatomic ions. The atomic mass of calcium, hydrogen, and oxygen is 40g, 1 g, and 16 respectively. Some nonmetals form a series of polyatomic ions with oxygen (all having the same. 
Given, the molecular formula of the given compound is Ca(OH)₂. (A cation is a positively charged ion an anion is a negatively charged ion.). 1 You can see that the outermost orbit of Oxygen has 6 electrons. To fill up the valence shell, which (when in oxygens row) has 8 electrons, an oxygen atom wants. Since each copper ion has a +1 charge and each oxygen ion has a -2 charge, we need two copper(I) ions to cancel out one oxygen ion. Now that two oppositely charged ions have formed, strong electrostatic. Then, the percentage of the elements by dividing the total mass of the element or or group of atoms by the molar mass of the compound multiplied by 100. Explanation : Oxygen has 6 valence electrons. Researchers at TU Wien (Vienna) have developed a groundbreaking oxygen-ion battery, which boasts exceptional durability, eliminates the need for rare elements, and solves the problem of fire hazards. Therefore, they readily take up these two electrons to form negatively charged O2- ions. Each electron carries a charge of 1 whereas each proton within the atomic nucleus carries a. Why does oxygen form O2 as an ion and not O or o3 - An atom either loses or gains one or more electrons to form an ion. 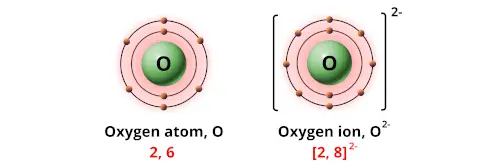
The percentage of an element can be calculated in 2 steps:įirst, calculate the molar mass of the compound by the addition of the atomic masses of the atoms. 2 Since every electron has a single negative charge, the addition of two electrons results in an oxygen ion with a charge of 2. 
The percentage of any element or group of atoms in a given compound is the number of parts by mass of that element or group of atoms present in 100 parts by mass of the given compound. You must determine the relative numbers of ions by balancing the positive and negative charges.The mass percentage of the OH⁻ in the compound Ca(OH)₂ is 45.94 %. Oxygen has six valence electrons, which are the electrons in the outer shell of the electron. The oxidation state of a simple ion like hydride is equal to the charge on the ion - in this case, -1. The compound’s name does not indicate that there are two nitrate ions for every barium ion. The ion name for oxygen is oxide.The charge of an oxygen ion is -2. Ions made from alkaline earth metals, the second group on the periodic table, have a 2+ charge. In this, the hydrogen is present as a hydride ion, H.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
